|
Cardiac grafts have been constructed using the new
technology of cell sheet engineering. This newest
generation of tissue engineering's based on the first
generation of tissue engineering, which used biodegradable
polymers to make scaffolds into which cells were inserted
to make tissue in the presence of growth factors.
First generation tissue engineering can be used for
constructing cartilage, bone, blood vessel, skin and
bladder tissue, which are amorphous or simple structures
that function physically and have a low blood supply
requirement and have large amounts of extracellular
matrix (ECM).
The second generation of tissue engineering is focusing
on tissue of the heart, liver, kidney, lung and brain
and will require different technology for their construction.
Micrometer-scale substructures, such as liver lobule
and nephrons, comprise these tissues, which have metabolic
functions, a large number of cells, less ECM, and
require more blood cells. Using scaffolds does not
allow for controlling the balance between ECM and
cells. Hence, a new method of tissue reconstruction
not based on scaffolds is needed, to avoid the fibrosis
that can occur in the heart, liver and kidney due
to excess ECM deposition.
|
|
|
Cell sheet engineering without scaffolds is an alternative
approach to tissue engineering. First, confluent cells
sheets are made on the culture dish. The harvesting
of these cell sheets allows for making layered, 3-dimensional
(3-D) structures.
Cell sheet construction involves the use of "intelligent
biomaterial" that is a temperature responsive
culture surface using a temperature responsive polymer
Poly(N-isopropylacrylamide) (PIPAAm). The cell sheet
is harvested by lowering the temperature. PIPAAm is
hydrophilic below 32 degrees Celsius and hydrophobic
above this temperature. Irradiation of PIPAAm with
an electron beam creates a surface that is slightly
hydrophobic and cell adhesive at 37 degrees Celsius,
and that become reversibly hydrophilic and non-cell
adhesive below 32 degrees Celsius due to rapid hydration
and swelling of grafted PIPAAm. This unique surface
change provides for spontaneous detachment of cultured
cells from the grafted surface by simply changing
the temperature. The use of trypsinization or another
enzyme disrupts the adhesive proteins and membrane
receptor ligand, while these are not disrupted when
reducing the temperature. Non-enzymatic cell sheet
harvest at 20 degrees Celsius allows recovery of the
cell sheet with good preservation of the cell surface
junction. Fibronectin, an ECM adhesive protein, completely
detaches from the surface. Hence, a good cell sheet
maintaining junction proteins can be made, which has
one that is sticky for adhesiveness.
|
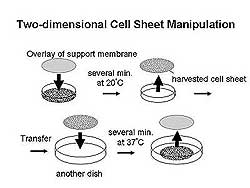 |
| Figure
1. Schematic of two-dimensional cell sheet manipulation. |
| Click
to enlarge |
|
Cardiomyocyte cell sheets constructed from chicken
embryo beat strongly after being detached from the
surface. Two-dimensional cell sheet manipulation involves
lowering the temperature to 20 degrees Celsius to
allow harvesting the cell sheet, which is then transferred
to another dish where the temperature is raised to
37% for several minutes causing the cell sheet to
adhere to the new surface after which the support
membrane can be removed (Figure 1). Using this system
it is possible to maintain the size and shape of cell
sheets that are transferred.
|
PAGE
TOP
|
Clinical Application of Engineered Cell Sheets |
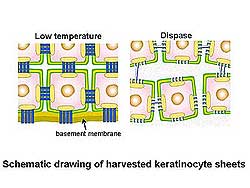 |
| Figure
2. Cultured keratinocyte sheets using 3T3 fibroblasts
were made and harvested using the dispase treatment
method. |
| Click
to enlarge |
|
Okano and colleagues are making single cell sheets
of skin, retina and corneal (epithelial, endothelial)
cells; homotypically layered multiple sheets of cardiac
muscle; and heterotypically layered multiple sheets
of liver, kidney and lung.
Cultured keratinocyte sheets using 3T3 fibroblasts
to be used as an artificial epidermis were made and
harvested by the dispase treatment method. However,
shortcomings using dispase for harvesting keratinocyte
sheets include a low-take ratio on deep wound and
susceptibility to infection. The low temperature method
maintains the cell junction and basement membrane,
while the dispase method does not (Figure 2). Corneal
transplantation with epithelial keratinocyte sheets
made using the low temperature method has been performed.
Polarized retinal pigmented epithelial (RPE) cell
sheets with growth factor inserted under the PIPPAm-grafted
porous membrane from the chick have been successfully
transplanted to the rabbit. Double-layered co-culture
tissue provides for prolonged survival of hepatocytes,
for at least several months (Figure 3).
|
PAGE
TOP
|
Application
for Cardiac Tissue Engineering |
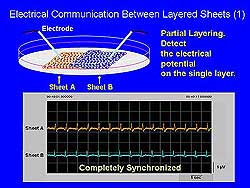 |
| Figure
4. Electrical communication between the two sheets
was synchronized, showing that adhesive proteins
maintained the structure and function between
layers. |
| Click
to enlarge |
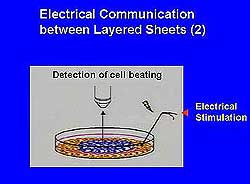 |
| Figure
5. The electrical stimulation was communicated
from one layer to the other. |
| Click
to enlarge |
|
The establishment of structural and functional communication
between the layered sheets is critical to constructing
cardiomyocyte cell sheets. Okano and colleagues constructed
primary cultured cardiomyocytes from neonatal rat
or chick embryos. Adhesive proteins maintained the
structure and function between the two layers and
electrical communication between the two sheets was
completely synchronized (Figure 4). Beating of the
cells was detected. Further, electrical stimulation
was transmitted from one sheet to the other (Figure
5). The bilayer sheets adhered closely without delamination.
Cross-sectional views showed connexin 43 present in
both tissue layers. When the cardiomyocyte sheets
were layered on to frame-like collagen membranes to
free them from rigid cell surfaces, synchronized movement
of both layers occurred. Communication and synchronized
movement with four-layer cell sheets on frame-like
collagen membranes with macroscopic beating was also
achieved. Four layer cardiac grafts were transplanted
into dorsal subcutaneous tissue of nude rats. At 3
weeks post-transplantation, beating of the cells was
seen on surface electrogram and skin resection revealed
continued beating of the cell sheets and formation
of new blood vessels. Histological analysis revealed
a large amount of neovascularization in the cardiac
grafts and connexin 43.
Cardiac grafts were transplanted into impaired
heart due to ischemic heart disease (IHD) or cardiomyopathy
(CM), in collaboration with Matsuda and Sawa at Osaka
University. Beating layered cardiomyocyte sheets were
transplanted onto the impaired heart after myocardial
infarction (MI), to determine whether the cell sheet
would adhere to the beating heart. Improvement in
cardiac performance in infarct rat hearts was seen
after transplantation of a 4-layer cardiomyocyte sheet.
The ejection fraction in the control animals was 45,
and increased to 64 in the transplanted animals.
|
PAGE
TOP
|
Report
Index | Previous Report
| Next Report
Scientific
Sessions | Activities
| Publications
Index
Copyright © 2002
Japanese Circulation Society
All Rights Reserved.
webmaster@j-circ.or.jp
|